Trending...
- Revenue Optics Expands Its Private Equity Practice as Sponsors Move Inside Sales to the Center of Distribution Value Creation
- Exclusive Red-Carpet Screening of High-Stakes Indie Thriller "Queen City: The Hornet's Nest" Coming to North Carolina on June 20th
- Nevada Boxing Hall of Fame Announces 14th Annual Induction Gala Weekend Honoring Classes of 2025 and 2026
NRx Pharmaceuticals (N A S D A Q: NRXP) $NRXP: Dual FDA Pathways, and an Expanding AI-Driven Treatment Ecosystem Position NRXP at the Forefront of a Multi-Billion-Dollar Mental Health Transformation
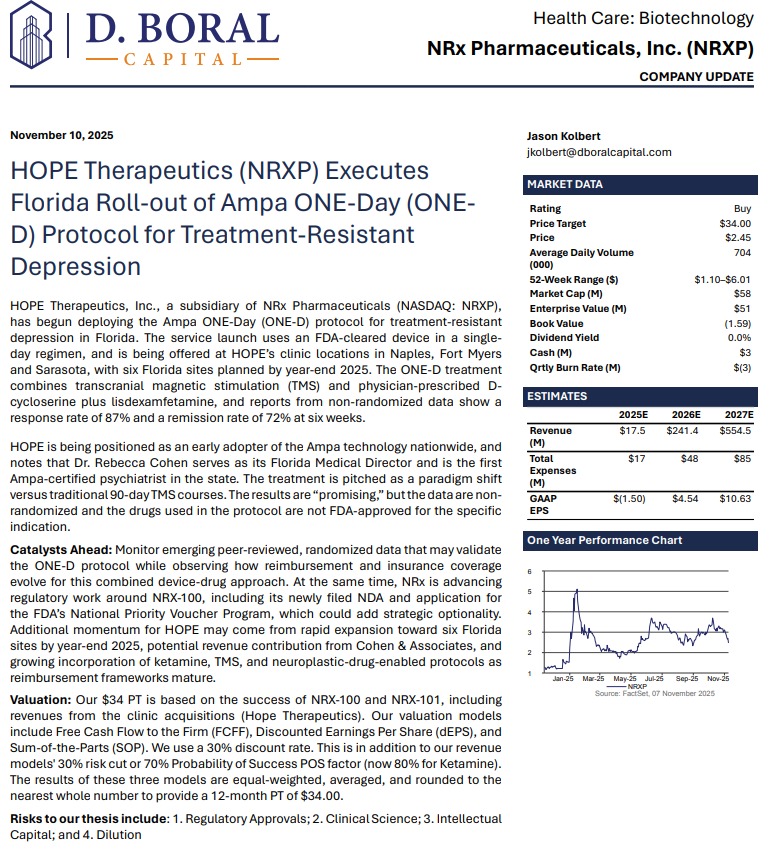
MIAMI - ncarol.com -- NRx Pharmaceuticals (N A S D A Q: NRXP) is rapidly emerging as a high-impact, late-stage biotech story transitioning into a nationally aligned healthcare platform, as a powerful convergence of regulatory clarity, federal policy support, and technological innovation reshapes both its timeline and total addressable market.
The company's latest developments place it squarely at the intersection of urgent public health need and government-backed acceleration, following the April 18, 2026 Presidential Executive Order aimed at fast-tracking treatments for serious mental illness, including psychedelic and breakthrough therapies.
This directive is expected to expand the use of real-world evidence, accelerate FDA review pathways, and introduce priority mechanisms such as Commissioner's National Priority Vouchers—one of which NRx has already applied for. The result is a meaningful shift in the regulatory landscape, positioning companies like NRx to benefit from faster approvals, increased prioritization, and national-level visibility.
FDA Alignment Transforms the Timeline
At the core of the investment thesis is a rare and highly material regulatory breakthrough. Following direct engagement with the FDA, NRx confirmed that its lead candidate, NRX-100 (preservative-free ketamine), can proceed toward a New Drug Application using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This positions the company to target an NDA submission as early as June 2026 while simultaneously expanding the proposed indication to include a broader population of patients suffering from treatment-resistant depression with suicidality.
To support this filing, NRx is preparing to submit data from more than 70,000 patients, creating one of the most robust real-world datasets in the psychiatric treatment space.
Key regulatory positioning now includes:
Dual FDA Catalysts Create a Rare Parallel Opportunity
More on ncarol.com
While the NDA pathway represents transformational upside, NRx is simultaneously advancing a second regulatory track that may generate nearer-term revenue.
The company's Abbreviated New Drug Application for preservative-free ketamine in anesthesia has already achieved bioequivalence confirmation and near-final labeling alignment, with anticipated FDA action in the second half of 2026.
This dual-track structure creates a compelling dynamic:
Few companies at this stage possess two aligned regulatory catalysts with overlapping timelines.
From Biotech to Platform: Expanding Into Neurotechnology and AI
NRx is no longer operating as a single-asset biotech. It is building an integrated platform that combines pharmaceuticals, neurostimulation, and artificial intelligence.
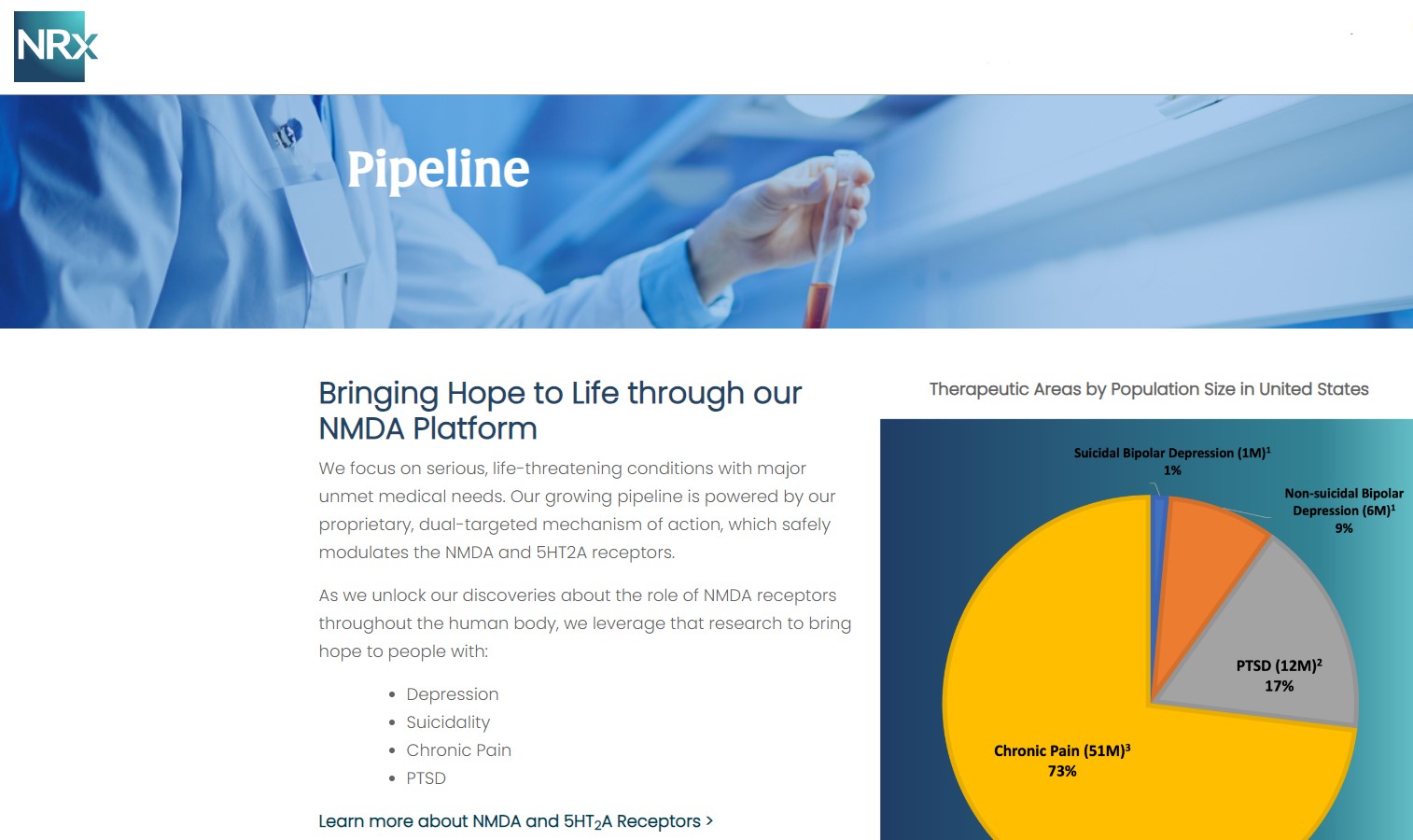
Its Breakthrough Therapy-designated candidate, NRX-101, is being developed as a maintenance therapy for suicidal bipolar depression and chronic pain, designed to work synergistically with neuroplastic treatment approaches.
That strategy is now expanding into advanced neurotechnology through the formation of NRx Defense Systems, a subsidiary focused on robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy.
This initiative targets military personnel and first responders—populations with significantly elevated rates of PTSD and depression—and introduces the potential for government-backed, non-dilutive funding channels.
Technology and platform expansion highlights:
Commercial Infrastructure Already in Motion
Importantly, NRx is not waiting for regulatory approvals to build its business. Through its HOPE Therapeutics subsidiary, the company has established a growing network of interventional psychiatry clinics delivering neuroplastic therapies, including ketamine and TMS.
These clinics are already generating revenue and are supported by a combination of government programs, insurance reimbursement, and private-pay patients.
In parallel, the company has partnered with neurocare AG to expand a nationwide treatment network that includes:
This infrastructure provides a ready-made commercialization channel upon approval.
More on ncarol.com
AI Integration Adds a New Layer of Precision Medicine
NRx is further differentiating itself through the integration of artificial intelligence into patient care.
Through its partnership with Emobot, the company is deploying a passive monitoring platform—often described as a "Depression Thermometer"—that continuously evaluates patient condition via smartphone-based data collection.
This enables earlier intervention, improved treatment optimization, and a scalable model for managing chronic mental health conditions.
Financial Position Strengthens Execution Capability
The company has also made meaningful progress in strengthening its financial foundation.
Recent disclosures highlight reduced operating expenses, the elimination of balance sheet debt, and sufficient capital to support operations through 2026.
This positions NRx to execute on its regulatory and commercial strategy without immediate financing pressure.
A Market Defined by Urgency and Scale
NRx is targeting one of the most significant unmet needs in healthcare. More than 13 million Americans report seriously considering suicide each year, while treatment-resistant depression and PTSD continue to rise globally.
With ketamine-based therapies demonstrating rapid clinical impact and federal policy now accelerating access to innovative treatments, the demand environment is both immediate and expanding.
Final Take: A Convergence Story with Multiple Inflection Points
NRx Pharmaceuticals is no longer simply advancing a drug candidate—it is building a multi-dimensional platform at the intersection of biotech, AI, neurotechnology, and national healthcare policy.
With:
NRx now represents a rare type of opportunity in the market:
A company where regulatory clarity, government support, and commercial execution are all advancing at the same time
For aggressive, forward-looking investors, NRXP is not just approaching a catalyst—
$NRXP is entering a phase where multiple catalysts are beginning to compound.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The company's latest developments place it squarely at the intersection of urgent public health need and government-backed acceleration, following the April 18, 2026 Presidential Executive Order aimed at fast-tracking treatments for serious mental illness, including psychedelic and breakthrough therapies.
This directive is expected to expand the use of real-world evidence, accelerate FDA review pathways, and introduce priority mechanisms such as Commissioner's National Priority Vouchers—one of which NRx has already applied for. The result is a meaningful shift in the regulatory landscape, positioning companies like NRx to benefit from faster approvals, increased prioritization, and national-level visibility.
FDA Alignment Transforms the Timeline
At the core of the investment thesis is a rare and highly material regulatory breakthrough. Following direct engagement with the FDA, NRx confirmed that its lead candidate, NRX-100 (preservative-free ketamine), can proceed toward a New Drug Application using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This positions the company to target an NDA submission as early as June 2026 while simultaneously expanding the proposed indication to include a broader population of patients suffering from treatment-resistant depression with suicidality.
To support this filing, NRx is preparing to submit data from more than 70,000 patients, creating one of the most robust real-world datasets in the psychiatric treatment space.
Key regulatory positioning now includes:
- NDA pathway for NRX-100 with no additional trials required
- Fast Track designation already in place
- Real-world evidence accepted as part of the approval framework
- Expanded label strategy significantly increasing market potential
Dual FDA Catalysts Create a Rare Parallel Opportunity
More on ncarol.com
- A Foundational Claim in Human Secrecy Goes Public
- Kappa Foundation of Charlotte Launches Transformational Community Investment Project
- Agape Leadership Academy Opens Nationwide Enrollment — State ESA Scholarships Cover Full Tuition for Families in 7 States
- Las Vegas Headliner Don Barnhart Brings National Touring Comedy Show to Comedy Cabana
- Nevada Boxing Hall of Fame Announces 14th Annual Induction Gala Weekend Honoring Classes of 2025 and 2026
While the NDA pathway represents transformational upside, NRx is simultaneously advancing a second regulatory track that may generate nearer-term revenue.
The company's Abbreviated New Drug Application for preservative-free ketamine in anesthesia has already achieved bioequivalence confirmation and near-final labeling alignment, with anticipated FDA action in the second half of 2026.
This dual-track structure creates a compelling dynamic:
- Near-term commercialization potential via ANDA approval
- Longer-term, high-value expansion via NDA approval
Few companies at this stage possess two aligned regulatory catalysts with overlapping timelines.
From Biotech to Platform: Expanding Into Neurotechnology and AI
NRx is no longer operating as a single-asset biotech. It is building an integrated platform that combines pharmaceuticals, neurostimulation, and artificial intelligence.
Its Breakthrough Therapy-designated candidate, NRX-101, is being developed as a maintenance therapy for suicidal bipolar depression and chronic pain, designed to work synergistically with neuroplastic treatment approaches.
That strategy is now expanding into advanced neurotechnology through the formation of NRx Defense Systems, a subsidiary focused on robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy.
This initiative targets military personnel and first responders—populations with significantly elevated rates of PTSD and depression—and introduces the potential for government-backed, non-dilutive funding channels.
Technology and platform expansion highlights:
- Development of robotic, AI-guided TMS systems
- Integration with NRX-101 for enhanced neuroplastic response
- Prototype expected mid-2026
- Clinical data showing up to 87% response rates in related therapies
Commercial Infrastructure Already in Motion
Importantly, NRx is not waiting for regulatory approvals to build its business. Through its HOPE Therapeutics subsidiary, the company has established a growing network of interventional psychiatry clinics delivering neuroplastic therapies, including ketamine and TMS.
These clinics are already generating revenue and are supported by a combination of government programs, insurance reimbursement, and private-pay patients.
In parallel, the company has partnered with neurocare AG to expand a nationwide treatment network that includes:
- 20+ clinical sites
- More than 400 installed TMS systems
This infrastructure provides a ready-made commercialization channel upon approval.
More on ncarol.com
- Brosix Celebrates 20 Years of Private Team Messaging for Small and Mid-Sized Businesses
- Top 15 Mosquito-Infested Cities in Louisiana and East Texas Ranked for 2026 Mosquito Season
- From Broken to Soaring Week 40
- Finnish Political Satire Film Generates 10,000+ Cross-Platform Interactions Following Gandalf Parody Video Across TikTok, YouTube and Telegram
- AI Is Making It Easier for API-First Platforms to Connect, Partner, Reach Customers, and Grow Revenue Faster
AI Integration Adds a New Layer of Precision Medicine
NRx is further differentiating itself through the integration of artificial intelligence into patient care.
Through its partnership with Emobot, the company is deploying a passive monitoring platform—often described as a "Depression Thermometer"—that continuously evaluates patient condition via smartphone-based data collection.
This enables earlier intervention, improved treatment optimization, and a scalable model for managing chronic mental health conditions.
Financial Position Strengthens Execution Capability
The company has also made meaningful progress in strengthening its financial foundation.
Recent disclosures highlight reduced operating expenses, the elimination of balance sheet debt, and sufficient capital to support operations through 2026.
This positions NRx to execute on its regulatory and commercial strategy without immediate financing pressure.
A Market Defined by Urgency and Scale
NRx is targeting one of the most significant unmet needs in healthcare. More than 13 million Americans report seriously considering suicide each year, while treatment-resistant depression and PTSD continue to rise globally.
With ketamine-based therapies demonstrating rapid clinical impact and federal policy now accelerating access to innovative treatments, the demand environment is both immediate and expanding.
Final Take: A Convergence Story with Multiple Inflection Points
NRx Pharmaceuticals is no longer simply advancing a drug candidate—it is building a multi-dimensional platform at the intersection of biotech, AI, neurotechnology, and national healthcare policy.
With:
- Federal acceleration of mental health treatment approvals
- FDA-aligned NDA pathway requiring no new trials
- Parallel ANDA approval opportunity
- Breakthrough Therapy designation for NRX-101
- Expanding clinic network generating real-world revenue
- AI-driven patient monitoring and treatment optimization
- Entry into defense-focused neurotechnology
NRx now represents a rare type of opportunity in the market:
A company where regulatory clarity, government support, and commercial execution are all advancing at the same time
For aggressive, forward-looking investors, NRXP is not just approaching a catalyst—
$NRXP is entering a phase where multiple catalysts are beginning to compound.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on ncarol.com
- Revenue Optics Expands Its Private Equity Practice as Sponsors Move Inside Sales to the Center of Distribution Value Creation
- Ecuador Freedom Launches First Scheduled Motorcycle Tour of Northern Peru's Lost Kingdoms
- New from Regal House Publishing, We Meet Apart, two sisters trapped in an Irish country manor
- Lineus Medical Completes Financial Restructuring with KMF Investments- Launching a New Era for SafeBreak
- Exclusive Red-Carpet Screening of High-Stakes Indie Thriller "Queen City: The Hornet's Nest" Coming to North Carolina on June 20th
- Neuro Recovery Institute Showcases Emerging Immersive Neuro-Rehabilitation Technology at Clinical Innovation Open House
- Community, Conservation & Waterwise Inspiration Bloom on June 6
- Industrial and systems engineers celebrate key leaders in the field at IISE Annual Conference
- Cosanostra Miami Rises as the Best Latin Nightclub in Miami in Under Two Years From its Opening
- CCHR Leader's 50-Year Fight for Psychiatric Drug Victims Gains National Momentum
- Author Releases 7-Day Screen Time Reset for Families as Teachers Worldwide Report Children "Struggling to Grasp Basic Concepts"
- Men's Health Month Begins with Record Proclamations, AP News Coverage, & National Momentum for Men's Health
- AdvisorVault Adds Social Media Archiving to its Consolidated D3P Service
- UK Financial Ltd Audits Full Ethereum Architecture Verifies Corporate Wallets and 19-Token Ecosystem Ahead of CoinMarketCap Filing for Global Ranking
- Creative Investment Research Analysis Finds Slower GDP Growth, Rising Inflation
- TechHouse Earns Highly Selective Microsoft Support Badge
- Chapel Hill Modernist Home Achieves Verified HERS Score of -29 in North Carolina's 100% Net-Zer
- J&J Exterminating Celebrates 65th Anniversary and Unveils Strategic Vision at Annual Team Meeting
- Tru by Hilton El Paso Airport Opens to Guests
- Zenylitics Announces Leadership Transition to Continue Accelerated Growth